[ad_1]
After receiving an experimental drug known as dostarlimab in a small trial, all 12 members skilled full remission. Docs say that is the primary time that such a most cancers drug has had a 100% success charge in a medical trial.
Outcomes of the small research, carried out by researchers on the Memorial Sloan Kettering Most cancers Middle in New York, had been revealed within the New England Journal of Drugs. The outcomes of the trial had been funded by the drug firm GlaxoSmithKline.
The most cancers sufferers acquired the drug intravenously each three weeks for six months.
Rectal most cancers therapy side-effects
Not one of the sufferers skilled a recurrence of their illness or wanted additional therapy over the course of a follow-up interval, which lasted round a yr on common, the research says.
Moreover, not one of the 12 sufferers skilled critical negative effects, which is without doubt one of the most important issues with typical rectal most cancers remedies.
The uncommon type of rectal most cancers examined within the trial usually doesn’t reply properly to chemotherapy
Though some had rashes, dermatitis, fatigue, pruritis or nausea, none skilled extra critical issues which can be widespread to rectal most cancers remedies — side-effects like infertility, neuropathy or sexual dysfunction.
Colorectal most cancers refers to colon and rectal most cancers
Typically each colon and rectal cancers are lumped collectively as “colorectal most cancers,” a grouping that represents the third most typical type of most cancers on this planet. However they don’t seem to be the identical factor.
Colon most cancers describes the existence of most cancers cells within the colon, whereas rectal most cancers describes most cancers within the rectum.
Rectal most cancers is much less widespread than colon most cancers and tougher to treatment. Frequent signs embrace rectal bleeding, constipation and belly ache.
The speed of remission is excessive if the illness is caught early.
When rectal most cancers is localized, the five-year survival charge is 90%. If it has unfold a bit bit, the speed lowers to 73% and if it is unfold quite a bit, the speed sinks to 17%.
Every of the sufferers on this latest research had a really particular type of rectal most cancers known as mismatch restore–poor rectal adenocarcinoma.
That type of the most cancers is even tougher to treatment than common rectal most cancers.
Mismatch restore–poor rectal most cancers is comparatively unusual — simply 5 to 10% of rectal most cancers sufferers expertise it and it would not reply properly to chemotherapy therapy.
Colorectal most cancers is a catchall time period for 2 kinds of most cancers which can be comparable, however not the identical
How important is the dostarlimab most cancers drug trial?
Dostarlimab will not be a brand new drug — it has been used to deal with endometrial most cancers.
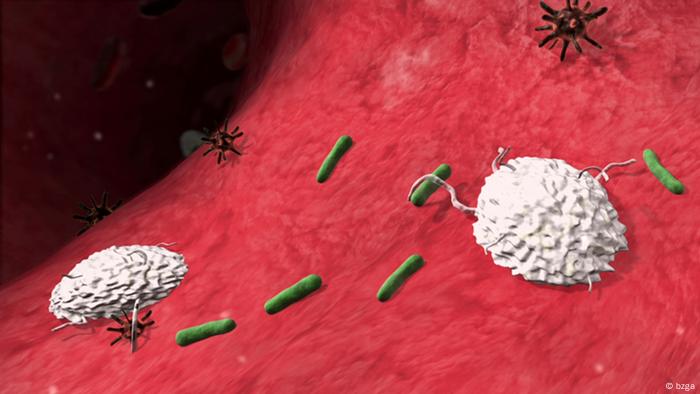
The drug is named a “checkpoint inhibitor,” which implies that as a substitute of killing most cancers cells outright, it permits an individual’s immune system to kill the cells by itself — it is due to this fact thought of an immunotherapy.
The outcomes are “trigger for nice optimism,” in response to Hannah Sanoff in an editorial revealed in the identical journal because the research outcomes. Sanoff is a most cancers physician and professor on the College of North Carolina at Chapel Hill. She was not concerned within the analysis.
Sanoff writes that the drug can’t but be used as an alternative to any present therapy for this or another type of most cancers.
The sufferers within the trial skilled what docs name a “medical full response” and so they have a greater prognosis than those that do not have a medical full response, however they may nonetheless see most cancers regrowth within the coming years, writes Sanoff.
“Little or no is thought concerning the period of time wanted to search out out whether or not a medical full response to dostarlimab equates to treatment,” Sanoff writes.

It’ll take time to know the extent to which immunotherapy can be utilized broadly to deal with varied types of most cancers
Too early to name it a most cancers treatment
Sanoff notes that it is also unclear whether or not the trial’s outcomes will likely be relevant to a extra common inhabitants of rectal most cancers sufferers, given the truth that all of the sufferers had a very uncommon type of the illness.
The research authors additionally observe the necessity to check the drug on extra folks earlier than they’ll attain any conclusion about its potential superiority to chemotherapy for treating rectal most cancers.
“The research is small and represents the expertise of a single establishment,” the authors write of their paper, including that the findings will must be reproduced in a bigger, extra racially and ethnically various group earlier than any choices about potential therapy could be made.
Sanoff writes that, given the caveats, the analysis presents solely “what could also be an early glimpse of a revolutionary therapy shift.”
Edited by: Zulfikar Abbany
[ad_2]
Source link